Florence H.J. van Tienen, Janneke G.J. Hoeijmakers, Christiaan van der Leij, Erika Timmer, Nikki Wanders, Patrick J. Lindsey, Fangzheng Yi, Fong Lin, Susanne P.M. Kortekaas, Helene Roelofs, Inge M. Westra, Pauline Meij, Lambert A.C.M. Wijnen, Irenaeus F.M. de Coo and Hubert J.M. Smeets.
Publication: Molecular Therapy Vol. 33 No 10 October 2025 pages 5061-5072
Abstract
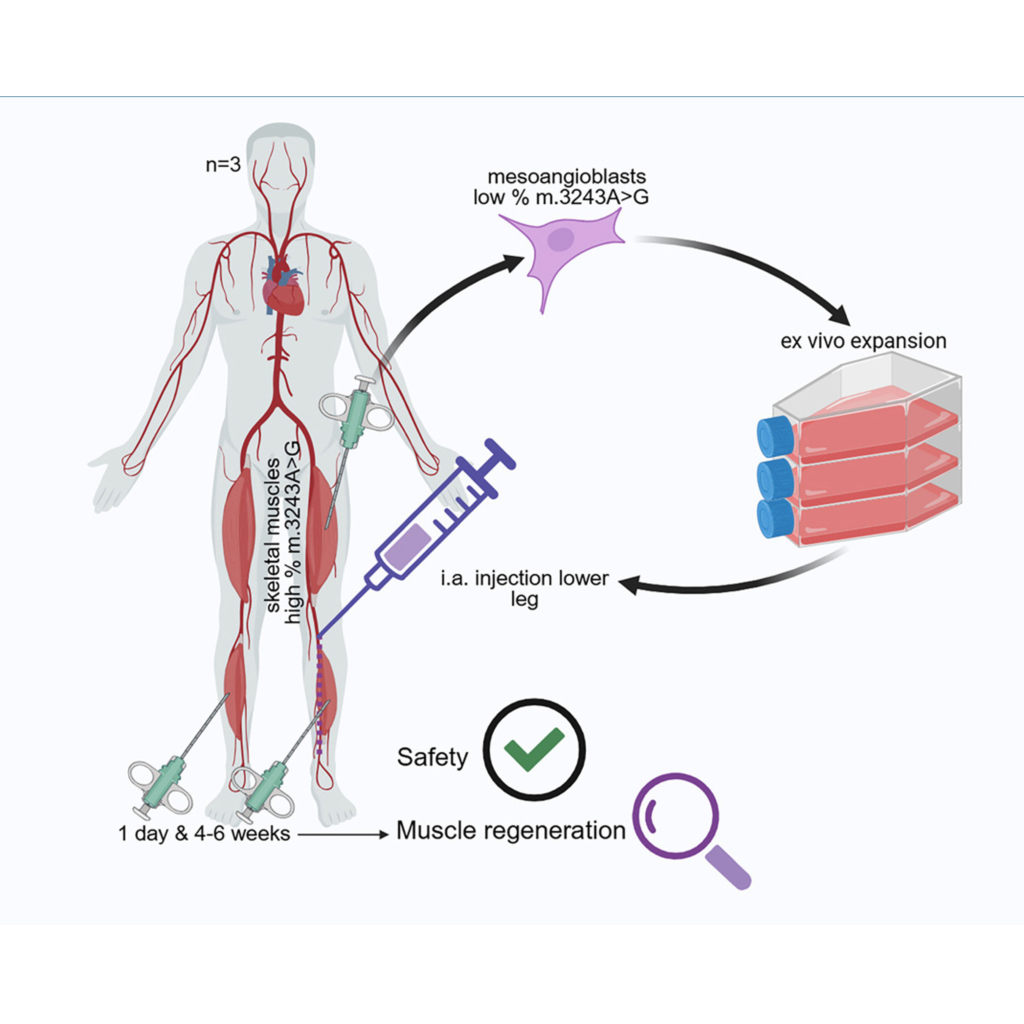
Intra-arterial transplantation of autologous mesoangioblasts in m.3243A>G mutation carriers is safe: First phase 1/2 human clinical study.
Progressive myopathy and exercise intolerance significantly impair quality of life in over 50% of m.3243A>G mutation carriers, with no curative therapy currently available. We hypothesize that intra-arterial administration of autologous, mtDNA mutation-free myogenic stem cells, mesoangioblasts, can reduce mutation load, enhance oxidative phosphorylation, and improve muscle function. To test this, the tibialis anterior muscles of three m.3243A>G mutation carriers were damaged by eccentric exercise before the infusion of 50 million/kg autologous mesoangioblasts into the left anterior tibial artery. The right tibialis anterior muscle served as control. Expanded mesoangioblasts had a mutation load of <15%, although culturing increased this by 7%–15%. Infusion caused mild, transient discomfort without serious adverse events or vascular obstructions, as confirmed by angiography. Blood and muscle biopsies revealed no systemic or local inflammation at 24 h and 4 weeks post-transplantation. Biopsies of the treated muscle suggested mesoangioblast migration and early signs of regeneration. This first-in-human study demonstrates that intra-arterial administration of autologous mesoangioblasts is safe, with promising, although inconclusive, evidence for muscle regeneration and mesoangioblast homing. These findings support further investigation into the therapeutic potential of mesoangioblasts for treating myopathy in m.3243A>G mutation carriers.
Read the article.